its not about consumption, the tank becomes normal after a specific time
Pressure-temperature law
***-Lussac's name is also associated — erroneously — with another gas law, the so-called pressure law, which states that:
Simply put, if a gas's temperature increases then so does its pressure, if the mass and volume of the gas are held constant. The law has a particularly simple mathematical form if the temperature is measured on an absolute scale, such as in
kelvins. The law can then be expressed mathematically as:
<DL><DD>
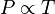
</DD></DL>or
<DL><DD>
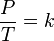
</DD></DL>where:
<DL><DD>
P is the
pressure of the gas.</DD><DD>
T is the
temperature of the gas (measured in Kelvin).</DD><DD>
k is a
constant.</DD></DL>This law holds true because temperature is a measure of the average
kinetic energy of a substance; as the kinetic energy of a gas increases, its particles collide with the container walls more rapidly, thereby exerting increased pressure.
For comparing the same substance under two different sets of conditions, the law can be written as:
<DL><DD>

</DD></DL>