@Fawi
I am not sure which whistling noise you are talking about, its been a long time since I saw the 2fast and 2furious movie. But I do remember FLi saying something about it.
@N2O
Well, 100% pure oxygen is very different from N2O. Nitrous oxide ( N2O ) contains 36 vol% oxygen, consequently the combustion chamber is filled with less useless nitrogen. It is also metered in as a liquid, which can cool the incoming charge further, thus effectively increasing the charge density. With all that oxygen, a lot more fuel can be squashed into the combustion chamber. The advantage of nitrous oxide is that it has a flame speed, when burned with hydrocarbon and alcohol fuels, that can be handled by current Internal Combustion engines, consequently the power is delivered in an orderly fashion, but rapidly. The same is not true for pure oxygen combustion with hydrocarbons, so leave that oxygen cylinder on the gas axe alone :-).
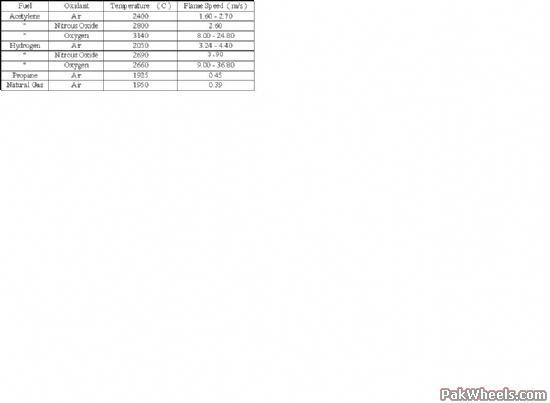
Essentially, pure oxygen would give more horsepower to a gasoline engine
because it is not diluted by nitrogen like it is in air - to a certain
point (RPM). The ignition timing of a gasoline engine is optimized for
running in diluted oxygen (air, which is mostly nitrogen). You would get
significantly better horsepower only at low RPM, where the burning time of
gasoline at 8 or more atmospheres is not critical. However, most engines
are designed to have optimized power in the 2500 to 4500 RPM range. At
high RPM, gasoline would burn too quickly and you would loose power. The
ignition characteristcs of the engine would have to be completely
re-designed and re-thought out from scratch for burning in pure oxygen.
Other oxidizers, like nitrous oxide, supply roughly the same ratio of
nitrogen to oxygen but are more reactive than air, which is why nitrous oxide supplies so much more power at high RPM - i.e., the burning rate is unchanged but the ability to supply oxygen on demand is better than with air.
Oxygen accelerates the combustion far too rapidly to be useful in an engine. The important thing to understand about nitrous is that it releases oxygen in a controlled manner during combustion. The disassociation of the N2O molecule absorbs energy and the nitrogen absorbs additional energy. All these moderate the effect of the oxygen release.
I just hope I have not confused you folks further coz I myself have very little knowledge about this but I am learning andhope you do the same.