What is a Catalytic Converter and What Does it do?
A catalytic converter is an emission control device that reduces emissions from an internal combustion engine. Since the 1970s governments around the world have been urging automobile manufacturers to build vehicles with low emissions. Catalytic Converters were introduced in American cars in 1975. Cars without catalytic converters release hydrocarbons, carbon monoxide and nitrogen oxide. These gases cause smog and are harmful to plants and health. Catalytic converters can also be found in buses, trucks, generators, and trains. In fact, almost every internal combustion engine has a form of catalytic converter attached to its exhaust. Every modern car is fitted with a catalytic converter.

What does a catalytic converter do?
The catalytic convert is found along the exhaust system of a vehicle. It looks like a small metal chamber. The catalytic converter receives exhaust gases and changes the chemical nature of the gases, to reduce the volume of toxic emissions from the car’s exhaust. The catalytic converter has a ceramic-based honeycomb structure that is lined with extremely precious metals; each metal has a specific job in emission-reduction.
The engine of a car produces three main emissions; Nitrogen Gas (N2). Carbon Dioxide (CO2) and Water Vapor (H2O). The catalytic converters tackle these emissions, which is why they are sometimes called three-way catalytic converters.
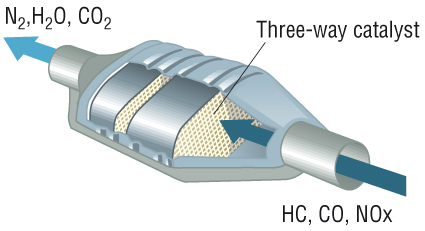
A catalyst is a substance that accelerates a chemical reaction without taking part in it. There are two types of catalysts found in a catalytic converter; the reduction catalyst and oxidization catalyst. The reduction catalyst uses platinum and rhodium which reduce NOx emissions. NOx is produced by nitrogen oxide and nitrogen dioxide which are released from the engine. The catalysts rip the nitrogen atoms from the molecules, therefore releasing cleaner oxygen.

The oxidization catalyst uses Platinum and Palladium to complete the job. These catalysts oxidize or burn the carbon monoxide and some hydrocarbons. This results in reduced amount of smog produced by evaporated unburnt fuel.
In the final stage of emission control involves O2 sensors. O2 sensors are found along the exhaust system. The O2 sensors detect the amount of oxygen found in the exhaust system and relay it to the ECU of the car. The on-board computer then adjusts the air/fuel ratio in order to allow engine to achieve the best conditions for an optimal chemical reaction. At this point, all the fuel entering the combustion chamber uses up all the oxygen provided in order to complete the combustion process.
The Disadvantage of a Catalytic Converter

In a sports car, the catalytic converter does this process more efficiently. The honeycomb structure in the catalytic converter has a few disadvantages to it. The first being that it slows down the gases leaving the exhaust system, which need to exit the system quickly and as efficiently as possible. This causes the engine to work harder as it struggles to breathe out the exhaust gases. The other disadvantage is that the honeycomb structure can fall apart by overheating, this blocks the exhaust system. Most people remove the catalytic converter, in order to get more power from the engine. Though this is not recommended. In many European countries and in the US, removing the catalytic converter is illegal.

This device is important as it greatly reduces the emissions of a car. All EURO II standard cars in Pakistan come along with a catalytic converter.



Hi friends, if anybody have a pick problem related from catalytic converter, please don’t remove the catalytic from your car, it will change the sound of your car, bring your car at our workshop we will clean the catalytic converter with german catalytic converter cleaner, total expense is 3000 rupees with labour including chemical, this process take just 30 minutes. yor car will give you excellent meilage and extra ordinary pick, smooth drive, like a zero meter car. after that process we will check your car at our scanner. our workshop Toyota Service Center and Spare parts is present near Islamabad airport chowk, new Gulzar-e- Quaid, contact us 0331-5600026. thanks
bring your car at out auto repair shop and get it clean call 35383616 .
Honey comb structure of my civic 2013 was damaged so i went to honda centre and they replaced it through Kharadia amd charged me 9000. Now it is working smoothly but sound of engine has increased. Pls. Advise